Warfarin: Difference between revisions
imported>Robert Badgett |
imported>Robert Badgett |
||
| Line 54: | Line 54: | ||
Because of warfarin's difficult [[pharmacokinetics]], researchers have proposed algorithms for warfarin loading. | Because of warfarin's difficult [[pharmacokinetics]], researchers have proposed algorithms for warfarin loading. | ||
{| class="wikitable" | {| class="wikitable" | ||
|+ [[Randomized controlled trial]]s of warfarin loading algorithms<ref name="pmid12729425">{{cite journal |author=Kovacs MJ, Rodger M, Anderson DR, ''et al'' |title=Comparison of 10-mg and 5-mg warfarin initiation nomograms together with low-molecular-weight heparin for outpatient treatment of acute venous thromboembolism. A randomized, double-blind, controlled trial |journal=Ann. Intern. Med. |volume=138 |issue=9 |pages=714-9 |year=2003 |pmid=12729425 |doi=|url=http://annals.org/cgi/content/full/138/9/714}}</ref><ref name="pmid9005747">{{cite journal |author=Harrison L, Johnston M, Massicotte MP, Crowther M, Moffat K, Hirsh J |title=Comparison of 5-mg and 10-mg loading doses in initiation of warfarin therapy |journal=Ann. Intern. Med. |volume=126 |issue=2 |pages=133–6 |year=1997 |pmid=9005747 |doi= |issn=|url=http://www.annals.org/cgi/content/full/126/2/133}}</ref><ref name="pmid9892329">{{cite journal |author=Crowther MA, Ginsberg JB, Kearon C, ''et al'' |title=A randomized trial comparing 5-mg and 10-mg warfarin loading doses |journal=Arch. Intern. Med. |volume=159 |issue=1 |pages=46–8 |year=1999 |pmid=9892329 |doi=|url=http://archinte.ama-assn.org/cgi/content/full/159/1/46}}</ref><ref name="pmid16893712">{{cite journal |author=Quiroz R, Gerhard-Herman M, Kosowsky JM, ''et al'' |title=Comparison of a single end point to determine optimal initial warfarin dosing (5 mg versus 10 mg) for venous thromboembolism |journal=Am. J. Cardiol. |volume=98 |issue=4 |pages=535–7 |year=2006 |pmid=16893712 |doi=10.1016/j.amjcard.2006.02.063}}</ref><ref name="pmid17989110">{{cite journal |author=Anderson JL, Horne BD, Stevens SM, ''et al'' |title=Randomized trial of genotype-guided versus standard warfarin dosing in patients initiating oral anticoagulation |journal=Circulation |volume=116 |issue=22 |pages=2563–70 |year=2007 |month=November |pmid=17989110 |doi=10.1161/CIRCULATIONAHA.107.737312 |url=http://circ.ahajournals.org/cgi/pmidlookup?view=long&pmid=17989110 |issn=}}</ref><ref name="pmid17851566">{{cite journal |author=Caraco Y, Blotnick S, Muszkat M |title=CYP2C9 genotype-guided warfarin prescribing enhances the efficacy and safety of anticoagulation: a prospective randomized controlled study |journal=Clin. Pharmacol. Ther. |volume=83 |issue=3 |pages=460–70 |year=2008 |month=March |pmid=17851566 |doi=10.1038/sj.clpt.6100316 |url=http://dx.doi.org/10.1038/sj.clpt.6100316 |issn=}}</ref> | |+ [[Randomized controlled trial]]s of warfarin loading algorithms<ref name="pmid12729425">{{cite journal |author=Kovacs MJ, Rodger M, Anderson DR, ''et al'' |title=Comparison of 10-mg and 5-mg warfarin initiation nomograms together with low-molecular-weight heparin for outpatient treatment of acute venous thromboembolism. A randomized, double-blind, controlled trial |journal=Ann. Intern. Med. |volume=138 |issue=9 |pages=714-9 |year=2003 |pmid=12729425 |doi=|url=http://annals.org/cgi/content/full/138/9/714}}</ref><ref name="pmid9005747">{{cite journal |author=Harrison L, Johnston M, Massicotte MP, Crowther M, Moffat K, Hirsh J |title=Comparison of 5-mg and 10-mg loading doses in initiation of warfarin therapy |journal=Ann. Intern. Med. |volume=126 |issue=2 |pages=133–6 |year=1997 |pmid=9005747 |doi= |issn=|url=http://www.annals.org/cgi/content/full/126/2/133}}</ref><ref name="pmid9892329">{{cite journal |author=Crowther MA, Ginsberg JB, Kearon C, ''et al'' |title=A randomized trial comparing 5-mg and 10-mg warfarin loading doses |journal=Arch. Intern. Med. |volume=159 |issue=1 |pages=46–8 |year=1999 |pmid=9892329 |doi=|url=http://archinte.ama-assn.org/cgi/content/full/159/1/46}}</ref><ref name="pmid16893712">{{cite journal |author=Quiroz R, Gerhard-Herman M, Kosowsky JM, ''et al'' |title=Comparison of a single end point to determine optimal initial warfarin dosing (5 mg versus 10 mg) for venous thromboembolism |journal=Am. J. Cardiol. |volume=98 |issue=4 |pages=535–7 |year=2006 |pmid=16893712 |doi=10.1016/j.amjcard.2006.02.063}}</ref><ref name="pmid17989110">{{cite journal |author=Anderson JL, Horne BD, Stevens SM, ''et al'' |title=Randomized trial of genotype-guided versus standard warfarin dosing in patients initiating oral anticoagulation |journal=Circulation |volume=116 |issue=22 |pages=2563–70 |year=2007 |month=November |pmid=17989110 |doi=10.1161/CIRCULATIONAHA.107.737312 |url=http://circ.ahajournals.org/cgi/pmidlookup?view=long&pmid=17989110 |issn=}}</ref><ref name="pmid17851566">{{cite journal |author=Caraco Y, Blotnick S, Muszkat M |title=CYP2C9 genotype-guided warfarin prescribing enhances the efficacy and safety of anticoagulation: a prospective randomized controlled study |journal=Clin. Pharmacol. Ther. |volume=83 |issue=3 |pages=460–70 |year=2008 |month=March |pmid=17851566 |doi=10.1038/sj.clpt.6100316 |url=http://dx.doi.org/10.1038/sj.clpt.6100316 |issn=}}</ref> | ||
! !! Dosing !!Study !! Time till a therapeutic INR<br/>(days)!! Rate of anticoagulation<br>(INR=2-3)!! Rate of over-coagulation | ! !! Dosing !!Study !! Time till a therapeutic INR<br/>(days)!! Rate of anticoagulation<br>(INR=2-3)!! Rate of over-coagulation | ||
|- | |- | ||
Revision as of 23:14, 23 February 2009
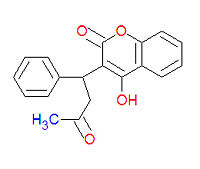
Warfarin (IUPAC name 4-hydroxy-3-(3-oxo-1-phenylbutyl)-2H-chromen-2-one), also widely called coumadin, is a an anticoagulant medication used prophylactically to suppress the formation of embolism and thrombosis. It was originally designed to be a rat poison. It works as an anticogulant by suppressing the enzyme epoxide reductase in the liver, thereby suppresing the formation of the reduced form of vitamin K epoxide, which is needed for the synthesis of many coagulation factors. As a drug, it is often sold as the sodium salt of warfarin.
Discovery
The dangers of feeding livestock spoiled sweet clover hay were known in the 1920’s[1] and scientists at the University of Wisconsin-Madison were spearheading the problem on multiple fronts. R.A. Brink and W.K. Smith were attempting to breed a new variety of clover that was free of the toxic effect and Karl Paul Link's laboratory was attempting to isolate the killer compound. Farmers were already hurting due to the Great Depression and despite knowing they should not feed their livestock such hay, they could not afford to buy uncontaminated supplies. This disease that was affecting many livestock throughout the US became known as "sweet clover disease".[2]
The urgency of this work was punctuated in the winter of 1933 when Ed Carlson, a farmer from Deer Park, Wisconsin, came to Madison for help carrying a milk can of uncoagulated blood, a dead cow in his truck and a sample of the hay contaminated with the spoiled sweet clover. This spurred Link on and in 1940 he finally published the source of the hemorrhagic factor in sweet clover, 3,3'-methylene-bis[4-hyfroxycoumarin], known as coumarin.[3] The following year Link published the discovery of dicumarol, the oxidised form of coumarin that was present in the spoiled sweet clover; this became widely used as an oral anticoagulants for medical treatment.[4][5]
Given the death of cattle from hemorrhaging, Link began to realise that anticoagulants had potential to be rodenticides.
I had an intuitive feeling that this might be a good thing. A pretty bad thing for rats, but a good thing for humans. But the idea didn't come overnight. It came into my head and the heads of everyone in the lab over a period of years. [6]
Dicumarol turned out to be a poor rodenticide as it acted too slowly[7] but by 1948 Link had patented another coumarin derivative as a rodenticide. Research for both dicumarol and the new rodenticide was funded by the Wisconsin Alumni Research Foundation (WARF) and the brand name for these anticoagulating rat posions was coined 'Warfarin' after WARF. Later, the rodenticide was promoted for clinical applications under the brand name 'Coumadin'.
Mechanism of action
Warfarin therapy reduces the vitamin K dependent cofactors II, VII, IX, and X and the vitamin K dependent Protein C. The level of factor II is thought to most influence coagulation.[8][9] The levels of factor VII and Protein C fall the fastest after warfarin is started.[9] With the exception of Factor IX, these factors are from either the extrinsic pathway or the final common pathway.
The effect of warfarin is measured by the prothrombin time (or the International Normalized Ratio derived from the prothrombin time) although warfarin can also affect the partial thromboplastin time.[10][11]
Pharmacokinetics
Absorption
Distribution
Metabolism
Pharmacogenomics
Warfarin activity is determined partially by genetic factors. The American Food and Drug Administration "highlights the opportunity for healthcare providers to use genetic tests to improve their initial estimate of what is a reasonable warfarin dose for individual patients".[12]
VKORC1
Genetic polymorphisms in the vitamin K epoxide reductase complex 1 (VKORC1) gene explain 30% of the dose variation between patients[13]: particular mutations make VKORC1 less susceptible to suppression by warfarin[14] There are a main haplotypes that explain 25% of variation: low-dose haplotype group (A) and a high-dose haplotype group (B).[15] For the three combinations of the haplotypes, the mean daily maintenance dose of warfarin was:
- A/A: 2.7+/-0.2 mg
- A/B: 4.9+/-0.2 mg
- B/B: 6.2+/-0.3 mg
VKORC1 polymorphisms also explain why African Americans are relatively resistant to warfarin (higher proportion of group B haplotypes), while Asian Americans are more sensitive (higher proportion of group A haplotypes).[15]
CYP2C9
CYP2C9 is an isoenzyme of cytochrome P-450. Polymorphisms of CYP2C9 explain another 10% of variation in warfarin dosing[13], mainly among Caucasian patients as these variants are rare in African American and most Asian populations.[16] A meta-analysis of mainly Caucasian patients found[16]:
- CYP2C9*2 allele:
- present in 12.2% of patients
- mean reduction was in warfarin dose was 0.85 mg (17% reduction)
- relative bleeding risk was 1.91
- CYP2C9*3 allele:
- present in 7.9% of patients
- mean reduction was in warfarin dose was 1.92 mg (37% reduction)
- relative bleeding risk was 1.77
Excretion
Dosage
Loading regimens
Because of warfarin's difficult pharmacokinetics, researchers have proposed algorithms for warfarin loading.
| Dosing | Study | Time till a therapeutic INR (days) |
Rate of anticoagulation (INR=2-3) |
Rate of over-coagulation | |
|---|---|---|---|---|---|
| Kovacs 10 mg | 10 mg/day for two days then specified adjusted dosing |
Kovacs†[17] | 4.2 | 83% by day 5 | 9% within 4 weeks(INR>5) (most occurred after day 10) |
| Quiroz‡[19] | 5 (two consecutive INRs≥2) |
56% at 5 days | 0% within 5 days (INR>5) | ||
| Anderson‡[20] | 69% at 5 days | ||||
| Harrison 10 mg | 10 mg/day for one day then flexible adjusted dosing |
Harrison¶[9] | 63% at 3.5 days | 17% within 3.5 days (INR>4.8) | |
| Crowther¶[18] | 69% at 5 days | 0% within 5 days (INR>5) 24% within 5 days (INR>3) | |||
| Kovacs 5 mg | 5 mg/day for two days then specified adjusted dosing |
Kovacs†[17] | 5.6 | 46% by day 5 | 11% within 4 weeks (INR>5) (most occurred after day 10) |
| Harrison 5 mg | 5 mg/day for one day then flexible adjusted dosing |
Harrison¶ [9] | 80% at 3.5 days | 4% within 4.5 days (INR>4.8) | |
| Crowther¶[18] | 88% at 5 days | 3% within 5 days (INR>5) 7% within 5 days (INR>3) | |||
| Quiroz‡[19] | 5 (two consecutive INRs≥2) |
52% at 5 days | 0% within 5 days (INR>5) | ||
| Anderson pharmacogenetic-guided algorithm | Based on CYP2C9 and VKORC1 | Anderson†[20] | 70% at 5 days | ||
| Caraco pharmacogenetic-guided algorithm | Based on CYP2C9 | Caraco[21] | 4.8 | ||
| † Blinded study. ‡ Independent study not from original investigators. ¶ Same research group (Hamilton, Ontario). Notes:
| |||||
Empiric dosing
On first look, the evidence table suggests that the Harrison 5 mg algorithm from Hamilton is the chest combination of efficacy and safety; however, two independent studies (Kovacs[17] and Quiroz in 2006 from the Massachusetts General Hospital[19]) have not been able to replicate the results of Hamilton group of Harrison and Crowther. One explanation may be that the personnel in the Harrison study were more expert and the flexibility in the algorithm allowed expression of their expertise. If so, then perhaps equally expert health care providers should use the Harrison 5 mg algorithm while other personnel should use the Kovacs 10 mg algorithm. Considering that the expertise of the Massachusetts General Hospital could not replicate the Hamilton results, perhaps most providers should use the Kovacs 10 mg algorithm, at least for inpatients.
A systematic review of the randomized controlled trials done through 2003 of 5 mg versus the 10 mg concluded that the Kovacs 10 mg regimen is best.[8] This conclusion was largely based on the inability of the results of the Harrison 5 mg flexible algorithm to be replicated by Kovacs.
Clinical practice guidelines in 2004 by the American College of Chest Physicians concluded that either 5 or 10 loads are acceptable.[22] The guidelines also state "if treatment is not urgent (eg, chronic stable atrial fibrillation), warfarin administration, without concurrent heparin administration, can be commenced out-of-hospital with an anticipated maintenance dose of 4 to 5 mg per day."[22]
Since publication of the systematic review[8] and clinical practice guidelines[22], a nonblinded, randomized controlled trial by Quiroz[19] found no difference between algorithms, but also achieved less frequent anticoagulation. The reason is not clear as the Kovacs 10 mg algorithm is very specific on each dose of warfarin. The Quiroz trial is also unique in that all patients were receiving a fondaparinux bridge.
Additional algorithms:
- The Tait 5 mg regimen is for outpatient anticoagulation. Patients are given 5 mg of warfarin per day for 5 days and then the INR is checked on day 5 to determine further dosing. (summary)[23]
- The Fennerty 10 mg regimen is an older regimen that has not been studied in an randomized controlled trial.[24][25]
Pharmacogenetic guided dosing
- The Kovacs 10 mg algorithm performed similarly to a pharmacogenetic-guided algorithm using 3CYP2C9 and VKORC1 .[20]
- A pharmacogenetic-guided algorithm using only CYP2C9 compared to a local algorithm.[21]
- A pharmacogenetic-guided algorithm using 3CYP2C9 and VKORC1 outperformed an unvalidated, clinical algorithm in modeled results.[26]
- A pharmacogenetic-based model from a cohort of orthopedic patients using 3CYP2C9 and VKORC1 genotype results predicted 80% of the variation in warfarin doses. It is awaiting validation in larger populations and has not been reproduced in those who require warfarin for other indications.[27]
- A cost-benefit analysis concludes that pharmacogenetic testing is unlikely to be cost effective for most patients.[28]
Adjusting the maintenance dose
Recommendations by the American College of Chest Physicians[22] have been distilled into a table to help manage dose adjustments.[29]
If the goal INR is 2 to 3, then adjustments should be made when the INR is 1.7 or less or when 3.3 or greater.[30]
Discontinuation before procedures
Management of warfarin around invasive procedures has been reviewed.[31]
One regimen before coronary artery bypass grafting is to stop 6 days before and give 5 mg/day of vitamin K on the day of warfarin cessation and use low molecular weight heparin with last dose the night before surgery.[32]
Interactions and contraindications
Warfarin interacts with many medications. A proposed classifications of mechanisms is:[33]
- Interference with platelet function
- Injury to gastrointestinal mucosa
- Reduced synthesis of vitamin K by intestinal flora
- Interference with warfarin metabolism by cytochrome P-450 CYP2C9 isoenzyme.
- Interruption of the vitamin K cycle
Some foodstuffs have also been reported to interact with warfarin.[34]
Adverse effects
Patients taking anti-platelet agents
Patients taking aspirin, clopidogrel, or dipyridamole may be at higher risk of hemorrhage.[35]
Elderly patients
Patients aged 80 years or more may be especially susceptible to bleeding complications with a rate of 13 bleeds per 100 person-years.[36]
Patients with prior intracranial hemorrhage from warfarin
These patients are at high risk of bad outcomes regardless of whether anticoagulation is resumed.[37]
Patients with cancer
| No cancer | Cancer | ||||
|---|---|---|---|---|---|
| Stage I or II | Stage III | Stage IV | |||
| Major bleeding |
Events per 100 patient-years |
8.6 | 3.4 | 19.1 | 42.8 |
| Hazard ratio | 1 | 0.5 | 2.15 | 4.8 | |
| Recurrent VTE |
Events per 100 patient-years |
12.8 | 14.5 | 44.1 | 54.1 |
| Hazard ratio | 1 | 1.9 | 5.3 | 4.6 | |
| Adapted from Table 3 of Prandoni et al.[38] | |||||
Patients with cancer are more likely to have bleeding complications, especially if they have Stage III (regionally extensive) or IV (metastatic) cancer.[38] Regardless of the extent of cancer, the risk of bleeding was less than the risk of recurrent embolism and thromboembolism:
Unstable anticoagualation
Supplementing warfarin with 150 micrograms of vitamin k may reduce the frequency of unstable anticoaguation among patients who are difficult to anticoagulate.[39] This amount of vitamin k led to 16% increase in the warfarin dose from 3.8 mg per day to 4.4 mg per day after one week.
Antagonism and reversal
A detailed table on reversing warfarin are provided in clinical practice guidelines from the American College of Chest Physicians.[22] For patients with an International Normalized Ratio (INR) between 4.5 and 10.0, 1 mg of oral vitamin K is effective.[40] Vitamin K should not be given subcutaneously.[41]
References
- ↑ Douglas Wardrop and David Keeling (2008) The story of the discovery of heparin and warfarin British Journal of Haematology 141 (6), 757-763
- ↑ Duxbury, B.M. & Poller, L. (2001) The oral anticoagulant saga: past, present, and future. Clinical and Applied Thrombosis/Hemostasis, 7, 269–275. PMID 11697707
- ↑ Campbell, H.A., Roberts, W.L., Smith, W.K. & Link, K.P. (1940) Studies on the hemorrhagic sweet clover disease. I. The preparation of hemorrhagic concentrates. Journal of Biological Chemistry, 136, 47–55.
- ↑ Campbell, H.A., Smith, W.K., Roberts, W.L. & Link, K.P. (1941) Studies on the hemorrhagic sweet clover disease. II. The bioassay of hemorrhagic concentrates by following the prothrombin level in the plasma of rabbit blood. Journal of Biological Chemistry, 138, 1–20.
- ↑ Stahmann, M.A., Huebner, C.F. & Link, K.P. (1941) Studies on the hemorrhagic sweet clover disease. V. Identification and synthesis of the hemorrhagic agent. Journal of Biological Chemistry, 138, 513–527.
- ↑ Karl Paul Link, Societal Contributions hosted by Wisconsin Alumni Research Foundation
- ↑ Last, J.A. (2002) The missing link: the story of Karl Paul Link. Toxicological Sciences, 66, 4–6.
- ↑ 8.0 8.1 8.2 8.3 Eckhoff CD, Didomenico RJ, Shapiro NL (2004). "Initiating warfarin therapy: 5 mg versus 10 mg". Ann Pharmacother 38 (12): 2115–21. DOI:10.1345/aph.1E083. PMID 15522981. Research Blogging.
- ↑ 9.0 9.1 9.2 9.3 9.4 Harrison L, Johnston M, Massicotte MP, Crowther M, Moffat K, Hirsh J (1997). "Comparison of 5-mg and 10-mg loading doses in initiation of warfarin therapy". Ann. Intern. Med. 126 (2): 133–6. PMID 9005747. [e]
- ↑ Bell DF, Harris WH, Kuter DJ, Wessinger SJ (1988). "Elevated partial thromboplastin time as an indicator of hemorrhagic risk in postoperative patients on warfarin prophylaxis". J Arthroplasty 3 (2): 181–4. PMID 3397749. [e]
- ↑ Hauser VM, Rozek SL (1986). "Effect of warfarin on the activated partial thromboplastin time". Drug Intell Clin Pharm 20 (12): 964–7. PMID 3816546. [e]
- ↑ FDA Approves Updated Warfarin (Coumadin) Prescribing Information. Retrieved on 2007-08-20.
- ↑ 13.0 13.1 Wadelius M, Chen LY, Downes K, et al (2005). "Common VKORC1 and GGCX polymorphisms associated with warfarin dose". Pharmacogenomics J. 5 (4): 262-70. DOI:10.1038/sj.tpj.6500313. PMID 15883587. Research Blogging.
- ↑ Rost S, Fregin A, Ivaskevicius V, et al (2004). "Mutations in VKORC1 cause warfarin resistance and multiple coagulation factor deficiency type 2". Nature 427 (6974): 537–41. DOI:10.1038/nature02214. PMID 14765194. Research Blogging.
- ↑ 15.0 15.1 Rieder MJ, Reiner AP, Gage BF, et al (2005). "Effect of VKORC1 haplotypes on transcriptional regulation and warfarin dose". N. Engl. J. Med. 352 (22): 2285-93. DOI:10.1056/NEJMoa044503. PMID 15930419. Research Blogging.
Cite error: Invalid
<ref>tag; name "pmid15930419" defined multiple times with different content - ↑ 16.0 16.1 Sanderson S, Emery J, Higgins J (2005). "CYP2C9 gene variants, drug dose, and bleeding risk in warfarin-treated patients: a HuGEnet systematic review and meta-analysis". Genet. Med. 7 (2): 97-104. PMID 15714076. [e]
- ↑ 17.0 17.1 17.2 17.3 Kovacs MJ, Rodger M, Anderson DR, et al (2003). "Comparison of 10-mg and 5-mg warfarin initiation nomograms together with low-molecular-weight heparin for outpatient treatment of acute venous thromboembolism. A randomized, double-blind, controlled trial". Ann. Intern. Med. 138 (9): 714-9. PMID 12729425. [e]
- ↑ 18.0 18.1 18.2 Crowther MA, Ginsberg JB, Kearon C, et al (1999). "A randomized trial comparing 5-mg and 10-mg warfarin loading doses". Arch. Intern. Med. 159 (1): 46–8. PMID 9892329. [e]
- ↑ 19.0 19.1 19.2 19.3 19.4 Quiroz R, Gerhard-Herman M, Kosowsky JM, et al (2006). "Comparison of a single end point to determine optimal initial warfarin dosing (5 mg versus 10 mg) for venous thromboembolism". Am. J. Cardiol. 98 (4): 535–7. DOI:10.1016/j.amjcard.2006.02.063. PMID 16893712. Research Blogging.
- ↑ 20.0 20.1 20.2 20.3 Anderson JL, Horne BD, Stevens SM, et al (November 2007). "Randomized trial of genotype-guided versus standard warfarin dosing in patients initiating oral anticoagulation". Circulation 116 (22): 2563–70. DOI:10.1161/CIRCULATIONAHA.107.737312. PMID 17989110. Research Blogging.
Cite error: Invalid
<ref>tag; name "pmid17989110" defined multiple times with different content - ↑ 21.0 21.1 21.2 Caraco Y, Blotnick S, Muszkat M (March 2008). "CYP2C9 genotype-guided warfarin prescribing enhances the efficacy and safety of anticoagulation: a prospective randomized controlled study". Clin. Pharmacol. Ther. 83 (3): 460–70. DOI:10.1038/sj.clpt.6100316. PMID 17851566. Research Blogging.
- ↑ 22.0 22.1 22.2 22.3 22.4 Ansell J, Hirsh J, Poller L, Bussey H, Jacobson A, Hylek E (2004). "The pharmacology and management of the vitamin K antagonists: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy". Chest 126 (3 Suppl): 204S–233S. DOI:10.1378/chest.126.3_suppl.204S. PMID 15383473. Research Blogging.
Cite error: Invalid
<ref>tag; name "pmid15383473" defined multiple times with different content Cite error: Invalid<ref>tag; name "pmid15383473" defined multiple times with different content - ↑ Tait RC, Sefcick A (1998). "A warfarin induction regimen for out-patient anticoagulation in patients with atrial fibrillation". Br. J. Haematol. 101 (3): 450-4. DOI:10.1046/j.1365-2141.1998.00716.x. PMID 9633885. Research Blogging.
- ↑ Fennerty A, Dolben J, Thomas P, et al (1984). "Flexible induction dose regimen for warfarin and prediction of maintenance dose". Br Med J (Clin Res Ed) 288 (6426): 1268–70. PMID 6424820. [e] [PubMed Central]
- ↑ Fennerty A, Campbell IA, Routledge PA (1988). "Anticoagulants in venous thromboembolism". BMJ 297 (6659): 1285-8. PMID 3144365. [e]
- ↑ Klein TE, Altman RB, Eriksson N, et al (February 2009). "Estimation of the warfarin dose with clinical and pharmacogenetic data". N. Engl. J. Med. 360 (8): 753–64. DOI:10.1056/NEJMoa0809329. PMID 19228618. Research Blogging.
- ↑ Millican E, Jacobsen-Lenzini PA, Milligan PE, et al (2007). "Genetic-based dosing in orthopaedic patients beginning warfarin therapy" 110 (5): 1511-5. DOI:10.1182/blood-2007-01-069609. PMID 17387222. Research Blogging. Online tool based on the study.
- ↑ http://www.annals.org/cgi/content/abstract/150/2/7
- ↑ Point-of-Care Guides - May 15, 2005 - American Family Physician. Retrieved on 2007-08-20.
- ↑ Rose AJ, Ozonoff A, Berlowitz DR, Henault LE, Hylek EM (January 2009). "Warfarin dose management affects INR control". J. Thromb. Haemost. 7 (1): 94–101. DOI:10.1111/j.1538-7836.2008.03199.x. PMID 18983486. Research Blogging.
- ↑ Kearon C, Hirsh J (May 1997). "Management of anticoagulation before and after elective surgery". N. Engl. J. Med. 336 (21): 1506–11. PMID 9154771. [e]
- ↑ Whitlock RP, Crowther MA, Warkentin TE, Blackall MH, Farrokhyar F, Teoh KH (2007). "Warfarin cessation before cardiopulmonary bypass: lessons learned from a randomized controlled trial of oral vitamin K". Ann. Thorac. Surg. 84 (1): 103–8. DOI:10.1016/j.athoracsur.2007.03.014. PMID 17588394. Research Blogging.
- ↑ Juurlink DN (August 2007). "Drug interactions with warfarin: what clinicians need to know". CMAJ : Canadian Medical Association journal = journal de l'Association medicale canadienne 177 (4): 369–71. DOI:10.1503/cmaj.070946. PMID 17698826. PMC 1942100. Research Blogging.
- ↑ Holbrook AM, Pereira JA, Labiris R, et al (2005). "Systematic overview of warfarin and its drug and food interactions". Arch. Intern. Med. 165 (10): 1095–106. DOI:10.1001/archinte.165.10.1095. PMID 15911722. Research Blogging.
- ↑ Johnson SG, Rogers K, Delate T, Witt DM (April 2008). "Outcomes associated with combined antiplatelet and anticoagulant therapy". Chest 133 (4): 948–54. DOI:10.1378/chest.07-2627. PMID 18198244. Research Blogging.
- ↑ Hylek EM, Evans-Molina C, Shea C, Henault LE, Regan S (2007). "Major hemorrhage and tolerability of warfarin in the first year of therapy among elderly patients with atrial fibrillation". Circulation 115 (21): 2689-96. DOI:10.1161/CIRCULATIONAHA.106.653048. PMID 17515465. Research Blogging. PMID 17515465
- ↑ Claassen DO, Kazemi N, Zubkov AY, Wijdicks EF, Rabinstein AA (October 2008). "Restarting anticoagulation therapy after warfarin-associated intracerebral hemorrhage". Arch. Neurol. 65 (10): 1313–8. DOI:10.1001/archneur.65.10.1313. PMID 18852344. Research Blogging.
- ↑ 38.0 38.1 Prandoni P, Lensing AW, Piccioli A, et al (2002). "Recurrent venous thromboembolism and bleeding complications during anticoagulant treatment in patients with cancer and venous thrombosis". Blood 100 (10): 3484–8. DOI:10.1182/blood-2002-01-0108. PMID 12393647. Research Blogging.
- ↑ Sconce E, Avery P, Wynne H, Kamali F (2007). "Vitamin K supplementation can improve stability of anticoagulation for patients with unexplained variability in response to warfarin". Blood 109 (6): 2419–23. DOI:10.1182/blood-2006-09-049262. PMID 17110451. Research Blogging.
- ↑ Crowther MA, Douketis JD, Schnurr T, et al (2002). "Oral vitamin K lowers the international normalized ratio more rapidly than subcutaneous vitamin K in the treatment of warfarin-associated coagulopathy. A randomized, controlled trial". Ann. Intern. Med. 137 (4): 251-4. PMID 12186515. [e]
- ↑ Dezee KJ, Shimeall WT, Douglas KM, Shumway NM, O'malley PG (2006). "Treatment of excessive anticoagulation with phytonadione (vitamin K): a meta-analysis". Arch. Intern. Med. 166 (4): 391–7. DOI:10.1001/.391. PMID 16505257. Research Blogging.