User:Milton Beychok/Sandbox: Difference between revisions
imported>Milton Beychok No edit summary |
imported>Milton Beychok No edit summary |
||
| Line 8: | Line 8: | ||
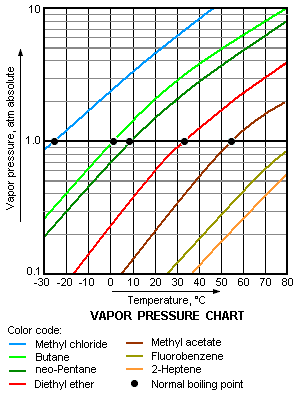
{{Image|Vapor Pressure Chart2.png|right|350px|A typical vapor pressure graph of various liquids.}} | {{Image|Vapor Pressure Chart2.png|right|350px|A typical vapor pressure graph of various liquids.}} | ||
== Vapor Pressure, temperature and boiling point == | |||
{{See also|Vapor pressure|boiling point|Antoine equation}} | |||
The vapor pressure of a substance is the pressure at which its gaseous (vapor) phase is in equilibrium with its liquid or solid phase. It is a measure of the tendency of [[molecule]]s and [[atom]]s to escape from a liquid or solid. A liquid's [[boiling point]] at [[atmospheric pressure]] corresponds to the temperature at which its vapor pressure is equal to the surrounding atmospheric pressure and is very commonly referred to as the ''normal boiling point''. | |||
Revision as of 20:13, 19 September 2010
In chemistry and physics, volatility is a term used to characterize the tendency of a substance to vaporize.[1] It is directly related to a substance' s vapor pressure. At a given temperature, a substance with a higher vapor pressure will vaporize more readily than a vapor with a lower vapor pressure.[2][3][4]
Any substance with a significant vapor pressure at temperatures of about 20 – 25 °C (68 – 77 °F) is very often referred to as being volatile.
In common usage, the term applies primarily to liquids. However, it may also be used to characterize the process of sublimation by which certain solid substances such as ammonium chloride (NH4Cl) and dry ice, which is solid carbon dioxide (CO2), change directly from their solid form to a vapor without becoming a liquid.
Any substance with a significant vapor pressure at temperatures of about 20 – 25 °C (68 – 77 °F) is very often referred to as being volatile.
Vapor Pressure, temperature and boiling point
- See also: Vapor pressure, boiling point, and Antoine equation
The vapor pressure of a substance is the pressure at which its gaseous (vapor) phase is in equilibrium with its liquid or solid phase. It is a measure of the tendency of molecules and atoms to escape from a liquid or solid. A liquid's boiling point at atmospheric pressure corresponds to the temperature at which its vapor pressure is equal to the surrounding atmospheric pressure and is very commonly referred to as the normal boiling point.
References
- ↑ Note: To vaporize means to become a vapor.
- ↑ Gases and Vapor (University of Kentucky website)
- ↑ James G. Speight (2006). The Chemistry and Technology of Petroleum, 4th Edition. CRC Press. ISBN 0-8493-9067-2.
- ↑ Kister, Henry Z. (1992). Distillation Design, 1st Edition. McGraw-hill. ISBN 0-07-034909-6.