Earth's atmosphere: Difference between revisions
imported>Milton Beychok m (→Pressure profile of the atmosphere: Corrected a CZ link) |
imported>Milton Beychok m (→Pressure profile of the atmosphere: Minor corrections) |
||
| Line 105: | Line 105: | ||
|- | |- | ||
|align=right|<math>M</math> | |align=right|<math>M</math> | ||
|align=left|= [[Molecular weight]] of air = 0. | |align=left|= [[Molecular weight]] of air = 0.028964 kg/[[Mole|mol]] | ||
|- | |- | ||
|align=right|<math>R</math> | |align=right|<math>R</math> | ||
| Line 111: | Line 111: | ||
|} | |} | ||
For example, the atmospheric pressure at an altitude of 10,000 metres is obtained as 26, | For example, the atmospheric pressure at an altitude of 10,000 metres is obtained as 26,437 Pascals by using Equation 1 and the appropriate base values for the altitude region number 1. | ||
{| border="0" width="400" align="right" cellpadding="0" cellspacing="0" style="wrap=no" | {| border="0" width="400" align="right" cellpadding="0" cellspacing="0" style="wrap=no" | ||
Revision as of 21:50, 18 August 2009

Atmospheric gases scatter blue light more than other wavelengths, giving the Earth a blue halo when seen from space at an altitude of 181 nautical miles (335 km) above the Earth.
The Earth's atmosphere is an envelope of gas that surrounds the Earth and extends from the Earth's surface out thousands of kilometres, becoming increasingly thinner (less dense) with distance but always held in place by Earth's gravitational pull. The atmosphere contains the air we breathe and it holds clouds of moisture (water vapor) that become the water we drink. It protects us from harmful solar radiation and warms the Earth's surface by heat retention. In effect, the atmosphere is an envelope that protects all life on Earth.
The atmosphere is a mixture of gases we call "air". On a dry volume basis, it consists of about 78% nitrogen and 21% oxygen. The remainder of about 1% contains argon, carbon dioxide and very small amounts of other gases. The atmosphere is rarely, if ever, completely dry. Water vapor is almost always present up to about 4% of the total volume. In desert regions, when dry winds are blowing, water vapor in the air will be nearly zero. This climbs in other regions to about 3% on extremely hot and humid days. The upper limit, approaching 4%, is for tropical areas.
The atmosphere has a total mass of about 5 × 1015 metric tons[1] and about 80% of that mass is within about 12 kilometres (7.5 miles) from the Earth's surface.
There is no definite boundary between the atmosphere and outer space. It slowly becomes thinner and fades into space.
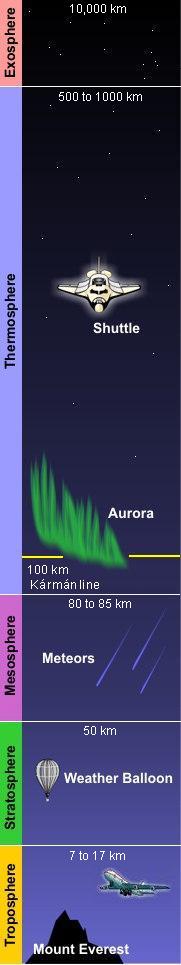
Structure of the atmosphere
As shown in the adjacent diagram, Earth's atmosphere has five primary layers, referred to as spheres. From the lowest to the highest layer, they are the Troposphere, Stratosphere, Mesosphere, Thermosphere and the Exosphere. The four boundaries between the primary layers, referred to as pauses, are the Tropopause, Stratopause, Mesopause and the Thermopause. In more detail:[2]
- Troposphere
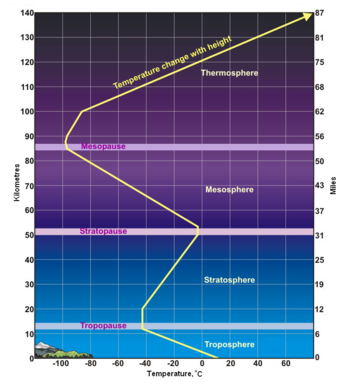
- From the Greek word "τρέπω" meaning to turn or change. The troposphere is the lowest layer of the atmosphere and extends from the Earth's surface to about 7 kilometres (4 miles) high at the north and south poles and 17 kilometres (11 miles) high at the equator. This is where we live. As the density of the air in this layer decrease with height, the air temperature in the troposphere also decreases with height. The temperature drops from about 15 to 16 °C at the surface[3][4][5][6] to about -45 °C at the top of the troposphere. All of our weather occurs in the troposphere. The troposphere contains about 80% of the total mass of the atmosphere. In fact, 50% percent of the total mass of the atmosphere is located in the lowest 5 to 6 kilometres (3.1 to 3.7 miles) of the troposphere.
- Atmospheric boundary layer
- The lowest part of the troposphere is called the atmospheric boundary layer (ABL) or the planetary boundary layer and extends from the Earth's surface to about 1.5 to 2.0 km in height. The air temperature of the atmospheric boundary layer decreases with increasing altitude until it reaches what is called the inversion layer (where the temperature increases with increasing altitude) that caps the atmospheric boundary layer. Above the inversion layer, the temperature decreases again.
- Tropopause
- The boundary between the troposphere and the stratosphere. Together, the troposphere and the tropopause are known as the lower atmosphere.
- Stratosphere
- From the Latin word "stratus" meaning spreading out. The stratosphere extends from the tropopause's height of 7 to 17 kilometres (4 to 11 miles) to a height of about 50 kilometres (31 miles) and contains about 19% of the atmospheric gases. The stratosphere experiences increasing temperature and solar radiation with height. It contains the ozone layer because the solar radiation is increasingly absorbed by oxygen molecules in the stratosphere, leading to the formation of ozone. The ozone layer resides in the lower portion of the stratosphere, though the thickness of the layer varies seasonally and geographically.
- Stratopause
- The boundary between the stratosphere and the mesosphere. The atmospheric pressure here is 1/1000th of the atmospheric pressure at sea level.
- Mesosphere
- From the Greek word "μέσος" meaning middle. The mesosphere extends from stratopause's height of about 50 kilometres (31 miles) to a height of about 80 to 85 kilometres (50 to 53 miles). The gases continue to become less and less dense with height. As such, the effect of the warming by ultraviolet radiation also becomes less and less, leading to a decrease in temperature with height. On average, temperature decreases to as low as -100 °C at the mesopause. However, the gases in the mesosphere are still dense enough to slow down meteors hurtling into the atmosphere, where they burn up and leave fiery trails in the night sky.
- Mesopause
- The boundary between the mesosphere and the thermosphere. It is the coldest place on Earth, with a temperature as low as -120 °C at times.
- Thermosphere
- The thermosphere extends from the mesopause's height of 80 to 85 kilometres (50 to 53 miles) to a height of between 500 and 1000 kilometres (310 and 620 miles) and is referred to as the upper atmosphere. The gases of the thermosphere are increasingly less dense than in the mesosphere. As such, incoming high energy ultraviolet and x-ray radiation from the sun, absorbed by the molecules in this layer, results in the temperature increasing to as high as 2000 °C near the top of this layer. However, despite the high temperature, this layer of the atmosphere would still feel very cold to our skin because of the extremely low density of the air. The total amount of energy from the very few molecules in this layer is not enough to heat our skin. The International Space Station orbits in this layer, between 320 and 380 kilometers (200 and 235 miles) from the Earth's surface. The Kármán line, named after the physicist Theodore von Kármán, is in the thermosphere at an altitude of 100 kilometres (62 miles) above the Earth and is often referred to as the boundary between Earth's atmosphere and outer space. [7]
- Thermopause
- The boundary between the thermosphere and the exosphere.
- Exosphere
- Extends from the thermopause's height of 500 to 1000 kilometres (310 to 620 miles) up to a height of 10,000 kilometres (6,200 miles) above the Earth's surface. The exosphere is the outermost layer of the atmosphere and it is in this layer that our man-made satellites orbit the Earth. From this layer, atoms and molecules escape into the void of space.
There are two other regions of the atmosphere that deserve discussion, one is the Ionosphere and the other is the Ozone layer:
- Ionosphere
- Solar radiation causes ionization of the molecules in a region of the atmosphere that extends from about 50 kilometers up to about 1100 kilometers above the Earth. That region is called the ionosphere and most of it is in the thermosphere, although it overlaps into the exosphere above the thermosphere and the mesosphere below the thermosphere. It is of importance because certain regions of the ionosphere make long distance radio communication possible by reflecting the radio waves back to Earth. It is also home to auroras. The lower edge of auroras occur at an altitude of about 100 kilometres (about the same height as the Kármán line) and may extend up to an altitude of 480 kilometres.[8]
- Ozone Layer
- Though part of the stratosphere, the physical and chemical composition of the ozone layer is far different from that of the stratosphere. Ozone (O3) in the Earth's stratosphere is created by ultraviolet light striking oxygen molecules containing two oxygen atoms (O2), splitting them into individual oxygen atoms (atomic oxygen). The atomic oxygen then combines with unbroken O2 to create O3, the ozone. Ozone is unstable (although long-lived in the stratosphere) and when ultraviolet light hits ozone, it splits into a molecule of O2 and an atom of atomic oxygen thus creating a continuing process called the ozone-oxygen cycle. This occurs in the ozone layer, the region from about 10 to 50 kilometres (6 to 31 miles) above Earth's surface. About 90% of the ozone in the Earth's atmosphere is contained in the stratosphere. Ozone concentrations are greatest between the heights of about 20 and 40 kilometeres above the Earth's surface, where they range from about 2 to 8 parts per million by volume.
|
Pressure profile of the atmosphere
Earth's atmospheric pressure at sea level is commonly taken to be 101,325 Pascals and it decreases with increasing altitude. There are two equations for calculating the atmospheric pressure at any given altitude up to 86 kilometres (53 miles). Equation 1 is used when the lapse rate is not equal to zero and equation 2 is used when the lapse rate equals zero:[9] The two equations are valid for seven different altitude regions of the Earth's atmosphere by using the designated base values (from the adjacent table) for , , and for each of the seven regions:[9]
- Equation 1:
- Equation 2:
| where: | |
| = Pressure at any given altitude , Pa | |
| = Base pressure, Pa | |
| = Base temperature, K | |
| = Base lapse rate, K/m | |
| = Base height (i.e., altitude), m | |
| = Any given altitude, m | |
| = Gravitational acceleration = 9.8067 m/s2 | |
| = Molecular weight of air = 0.028964 kg/mol | |
| = Universal gas constant = 8.3144 J/K mol) |
For example, the atmospheric pressure at an altitude of 10,000 metres is obtained as 26,437 Pascals by using Equation 1 and the appropriate base values for the altitude region number 1.
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Composition of the atmospheric air
The table below lists the concentration of 14 gases present in filtered dry air. Two of the gases, nitrogen and oxygen make up 99.03 percent of the clean, dry air. The other listed gases total to 0.97 percent.
Note the amounts of greenhouse gases that are present: water vapor, carbon dioxide, methane, nitrous oxide, and ozone. Additional gases (not listed in the table) are also present in very minute amounts.
The atmospheric air is rarely, if ever, dry. Water vapor is nearly always present up to about 4% of the total volume. In the deserts regions, when dry winds are blowing, the water vapor content will be near zero. This climbs to near 3% on extremely hot/humid days. The upper limit of 4% is for tropical climates.
Unfiltered air contains minute amounts of various types of particulate matter derived from sources such as from dust, pollen and spores, sea spray, volcanoes, meteoroids and industrial activities.
References
- ↑ The Mass of the Atmosphere: A Constraint on Global Analysis K.E. Trenberth and Lesley Smith, National Center for Atmospheric Research, Boulder, Colorado
- ↑ 2.0 2.1 2.2 The Layers of the Atmosphere From the website of the National Oceanic and Atmospheric Administration (NOAA) and the National Weather Service (NWS) The primary source for this section.
- ↑ Earth's Atmosphere
- ↑ NASA - Earth Fact Sheet
- ↑ Global Surface Temperature Anomalies
- ↑ Earth's Radiation Balance and Oceanic Heat Fluxes
- ↑ This definition is accepted by the Fédération Aéronautique Internationale (FAI), an international standard setting and record-keeping body for aeronautics and astronautics.
- ↑ Asahi Aurora Classroom Geophysical Institute, University of Alaska
- ↑ 9.0 9.1 U.S. Standard Atmosphere, 1976 Scroll to pdf page 28 of 241 pdf pages.
- ↑ The Atmosphere From the website of the National Oceanic and Atmospheric Administration (NOAA) and the National Weather Service (NWS), Last updated May 5, 2009




![{\displaystyle P=P_{b}\cdot \left[{\frac {T_{b}}{T_{b}+L_{b}\cdot (h-h_{b})}}\right]^{\textstyle {\frac {g\cdot M}{R\cdot L_{b}}}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/a0bf08544e1a140969dae6359b67f791120f5190)
![{\displaystyle P=P_{b}\cdot \exp \left[{\frac {-\,g\cdot M\cdot (h-h_{b})}{R\cdot T_{b}}}\right]}](https://wikimedia.org/api/rest_v1/media/math/render/svg/e59f45ff65284fa6d5051aed5644e4a9383a5745)




